Abstract
Background: Activating mutations in KIT are infrequently observed in myeloid neoplasms, but are a pathognomonic finding in systemic mastocytosis (SM) where the hotspot D816V variant is documented in 90-95% of patients. In SM, KIT variants may occur at a low variant allele frequency (VAF) below the limits of detection of most commercial next generation sequencing (NGS) assays necessitating digital droplet PCR (ddPCR) for accurate detection. Aggressive SM often presents concurrently with an associated hematologic neoplasm (SM-AHN). Chronic myelomonocytic leukemia (CMML) is the most frequently co-occurring hematologic malignancy, but KIT mutations are infrequently detected by conventional NGS. The limitations in detecting KIT variants by NGS and increased frequency of CMML in SM-AHN suggest that subclonal KIT mutations may be underrecognized in CMML. Further, identification may have prognostic and therapeutic implications given the emergence of KIT-targeted therapies in SM.
Methods: A multicenter, retrospective study was performed to assess the prevalence of subclonal KIT D816V mutations in CMML patients and examine clinical implications. Eligible patients had pathologically confirmed CMML by WHO 2017 criteria, with available bone marrow (n=59) or peripheral blood (n=14) sample obtained prior to therapy. Samples underwent DNA sequencing using a 285-gene NGS panel (QIAseq), with a VAF threshold of 5% utilized for variant calling. KIT D816V mutations were assessed by ddPCR, with limit of detection of 0.03%. Clinical data was collected from baseline and follow up for phenotypic and prognostic comparisons. Fishers exact and Chi-square tests were used to compare categorical variables and the Mann-Whitney test for continuous variables. The Kaplan-Meier method was used to estimate overall survival (OS), with the log rank test used to compare survival between groups. The study was approved by institutional review boards at each institution.
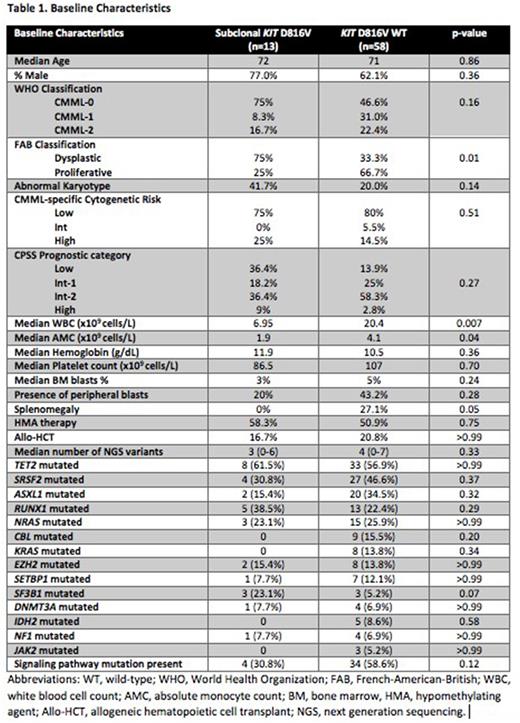
Results: A total of 73 patients were included with a median age of 71, and 66% were male. By WHO criteria, 50% were classified as CMML-0, 28% CMML-1, and 22% CMML-2; 59% of patients had proliferative CMML by FAB criteria. KIT D816V mutations were detected in 15 patients (21%) by ddPCR, compared to two (3%) by NGS. The median VAF of mutations detected by ddPCR was 0.06%, whereas the two variants detected by NGS had VAFs of 44.1% and 47.0%. Baseline characteristics of KIT wild-type (WT) and mutated (MUT) patients are detailed in Table 1. KIT MUT patients had lower white blood cell (p=0.007) and absolute monocyte counts (p=0.04), less frequent splenomegaly (p=0.05), more frequent karyotype abnormalities (p=0.14), and lower frequency of proliferative disease by FAB criteria (p=0.01).
There was a median of 4 variants (range 0-7) detected per patient by NGS, and the most frequently mutated genes were TET2 (59%), SRSF2 (44%), ASXL1 (33%),RUNX1 (25%), and NRAS (25%). KIT MUT patients had a median of 3 total variants (range 0-6) detected by NGS compared to 4 (range 0-7) in KIT WT (p=0.63). There was a trend toward higher frequency of SF3B1 (23% vs. 5%, p=0.07) mutations in KIT MUT patients. Conversely, no mutations were observed in KRAS or CBL in KIT MUT patients, compared to 14% and 16% in KIT WT, respectively (p=0.34, p=0.20). When evaluating mutations in signaling pathways, there was a trend toward lower frequency in KIT MUT patients compared to WT (31% vs. 59%, p=0.12).
With a median follow up of 48 months, median OS of the cohort was 30 months. KIT MUT patients demonstrated an OS of 27.5 months compared to 30 months in WT patients (HR 1.06, p=0.87). Transformation to AML was observed in 42% of KIT MUT patients compared to 29% of WT patients (p=0.49). A similar proportion of patients were treated with hypomethylating agents (HMAs), with a response rate of 14% in KIT MUT patients compared to 32% in WT (p=0.63), and complete remission rates of 0% vs. 27%, respectively.
Conclusions: This study is the first to document the prevalence of subclonal KIT D816V mutations by ddPCR in CMML, revealing a high prevalence in comparison to detection by standard NGS assays. While sample size limited statistical comparisons, KIT mutations were associated with distinct phenotypic features, including less proliferative disease and poor response to HMAs. Given the efficacy of KIT inhibitors in SM-CMML, these findings could represent an avenue for targeted therapy in CMML.
Disclosures
Geissler:Celgene: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria; Jazz Pharmaceuticals: Consultancy, Honoraria. Palomo:Janssen: Consultancy. Santini:AbbVie: Membership on an entity's Board of Directors or advisory committees; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees; Geron: Membership on an entity's Board of Directors or advisory committees; Menarini: Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees; Otsuka: Membership on an entity's Board of Directors or advisory committees; Servier: Membership on an entity's Board of Directors or advisory committees; Syros: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees. Graubert:H3 Biomedicine: Honoraria; Juno Therapeutics: Honoraria; Janssen: Research Funding. Thota:BMS: Membership on an entity's Board of Directors or advisory committees; Blueprint Medicine: Membership on an entity's Board of Directors or advisory committees. Griffiths:Astex Pharmaceuticals: Research Funding; BMS/Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Blueprint Medicines: Research Funding; Celldex Therapeutics: Research Funding; CTI Biopharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees; Medicom Worldwide: Honoraria; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Physician Educational Resource: Honoraria; Picnic Health: Honoraria; Takeda Oncology: Consultancy, Membership on an entity's Board of Directors or advisory committees; Taiho Oncology: Consultancy, Membership on an entity's Board of Directors or advisory committees; AstraZeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees; Apellis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Alexion: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees; AAMDSIF: Honoraria. Pleyer:AbbVie: Honoraria; BMS: Honoraria, Other: Travel/Education grant; Novartis: Honoraria, Other: Travel/education grant; Kite-Giliead: Other: Travel/Education grant; BeiGene: Other: Travel/Education grant. Thol:BMS: Membership on an entity's Board of Directors or advisory committees; AbbVie: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees. Bejar:Aptose Biosciences: Current Employment, Current equity holder in publicly-traded company; Gilead: Other: data safety monitoring committees chair; Epizyme: Other: data safety monitoring committee chair; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Research Funding. Sallman:Lixte: Patents & Royalties: LB-100; Agios: Membership on an entity's Board of Directors or advisory committees; Aprea: Membership on an entity's Board of Directors or advisory committees, Research Funding; Incyte: Speakers Bureau; Intellia: Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Magenta: Consultancy; Kite: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; AbbVie: Membership on an entity's Board of Directors or advisory committees; Shattuck Labs: Membership on an entity's Board of Directors or advisory committees; Syndax: Membership on an entity's Board of Directors or advisory committees; Syntrix Pharmaceuticals: Research Funding; Takeda: Consultancy; Nemucore: Membership on an entity's Board of Directors or advisory committees. Kuykendall:Pharmaessentia: Consultancy, Honoraria, Speakers Bureau; Imago Biosciences: Consultancy, Honoraria, Speakers Bureau; Incyte: Consultancy, Honoraria, Speakers Bureau; Blueprint: Consultancy, Honoraria, Speakers Bureau; Novartis: Consultancy, Honoraria, Speakers Bureau; Abbvie: Consultancy, Honoraria, Speakers Bureau; GSK - Sierra Oncology: Consultancy, Honoraria, Other: Research Support, Speakers Bureau; Prelude Pharmaceuticals: Other: Research Support; BMS: Consultancy, Honoraria, Other: Research Support, Speakers Bureau; Morphosys: Other: Research Support; Protagonist: Other: Research Support; CTI Biopharma: Consultancy, Honoraria, Speakers Bureau. Komrokji:Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Geron: Consultancy; Servier: Consultancy, Honoraria, Speakers Bureau; AbbVie: Consultancy, Honoraria, Speakers Bureau; Taiho Oncology: Consultancy, Membership on an entity's Board of Directors or advisory committees; Acceleron Pharma: Consultancy; PharmaEssentia, Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees; CTI BioPharma, Innovent: Honoraria, Membership on an entity's Board of Directors or advisory committees; Jazz Pharmaceuticals: Consultancy, Honoraria, Speakers Bureau; Bristol Myers Squibb: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau. George:Blueprint Medicines Corporation: Consultancy, Membership on an entity's Board of Directors or advisory committees; Incyte: Consultancy, Membership on an entity's Board of Directors or advisory committees; Celgene/BMS: Consultancy, Membership on an entity's Board of Directors or advisory committees; ARUP Laboratories: Other: Associated; Cogent Biosciences: Consultancy, Membership on an entity's Board of Directors or advisory committees. Padron:Blueprint: Honoraria; Kura: Research Funding; Incyte: Research Funding; Stemline: Honoraria; BMS: Research Funding; Taiho: Honoraria; Syntrix Pharmaceuticals: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.